More than 25 years experience in providing automation for tablet dissolution testing.
Automated Lab Systems provides state of the art laboratory testing equipment and complimentary products worldwide. We serve the Pharmaceutical, Environmental, Food and Sample Preparation industries among others through our international network of distributors.
Starting back in 1992 with our first automated Dissolution system based on a relational database using Btrieve, ALS has grown and expanded its portfolio of products and applications and now supplies high quality and extremely dependable applications for demanding tablet dissolution testing, sample preparation and dispensing using high end databases Oracle and MySQL for local and remote data storage.
Tablet Dissolution Testing
Reliable and fully compliant equipment and associated accessories are an extremely important part of being able to achieve consistency across all your testing and choosing your lab equipment carefully can improve the quality of your results. Our automated dissolution testing is unique in providing both UV and HPLC detection and integrates dissolution testers, UV Spectrophotometers and HPLC CDS from different manufacturers.
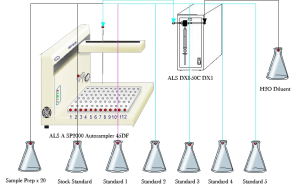
Sample Preparation
Sample preparation is one of the most time consuming and labour intensive areas of any laboratory. Automating this area can release valuable staff time to concentrate on the results rather than preparation. Our sample preparation system is a must for any testing laboratory. It offers flexibility, compatibility and infinitely variable parameters to enable the desired preparation.
Content Uniformity
Uniformity of Content and Assay is a pharmaceutical analysis technique for the quality control of hard shell gelatin capsules or tablets. The test for Content Uniformity is required for Coated Tablets, Trans-dermal systems, Suspensions, Inhalations, Solids and Suppositories etc. We automate this analysis with both UV_Visible and HPLC detection.

We are proud to have ISO 9001 certification, an internationally recognised standard that ensures our products and services are delivered consistently through an effective quality management system. This has been in place since 2013.
Our decision to work to ISO 9001 accreditation demonstrates our commitment to providing a high-quality and consistent service to our customers with ongoing investment in technology, development, processes and procedures.
The benefits of ISO 9001 go far beyond meeting customer requirements – management systems help to enhance professionalism of our businesses operations and provide a culture of exceeding expectations. This is helpful with employee engagement as they are clear on expectations and goals and jointly share in the success of business growth.
We are dedicated to constant improvement and development and achieving and maintaining an ISO 9001 accreditation ensures we have the processes and systems in place to do.